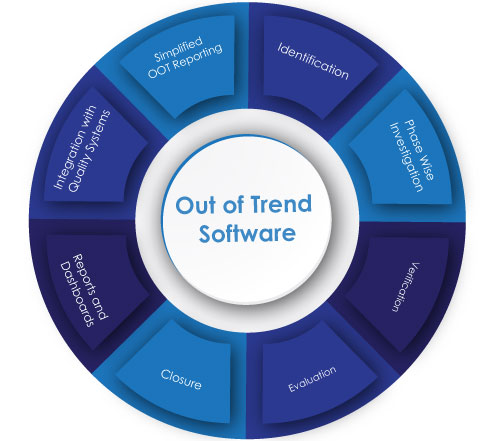
Out of Trend(OOT)
Out of Trend (OOT) events play a significant role in quality control and process management within the life sciences industry. An OOT event occurs when data points deviate from an expected pattern or trend, indicating a variation that could potentially impact product quality or process performance if left unaddressed. Unlike out-of-specification (OOS)
OOT deviations represent unusual fluctuations that require attention. However, it is important to note that OOT events still require compliance with regulatory standards such as 21 CFR Part 211 and 820 in order to investigate and resolve their potential causes effectively.
OOT Investigation
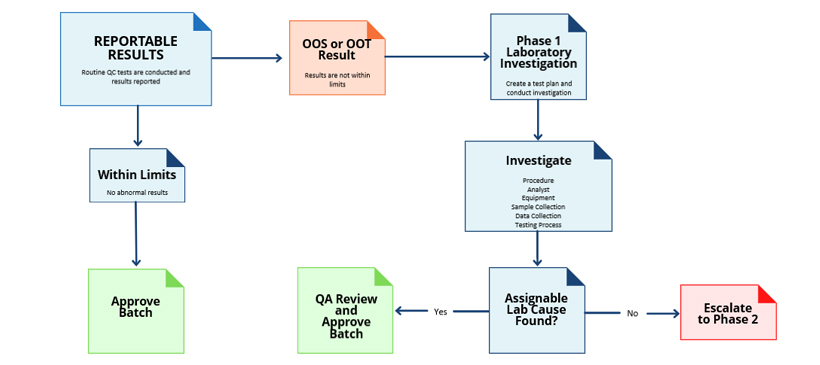
Once an OOT event is detected, a thorough investigation becomes critical to maintaining compliance and confidence in life science operations. The investigation typically involves several key steps. First and foremost, is performing a root cause analysis study to establish the causes of variation.
Variability in process, equipment, environment, or operations may contribute to the OOT event. Therefore, any process modifications made recently should be thoroughly assessed. Uncontrolled process or product changes often generate unexpected variations that mimic OOT or OOS results. Establishing control over all changes is key to avoiding false signals.
Statistical tools for OOT Investigation
Detecting an OOT event often relies on various statistical methods, each providing unique insights into the deviation and its implications. Most commonly used statistical tools for detecting an OOT event include Statistical Process Control (SPC) charts.
The SPC charts are effective tools for monitoring data over time and establishing an expected range of variability. Points outside the control limits on the chart indicate an OOT signal. Control charts play a crucial role in detecting changes in the process that could lead to nonconforming products even before specifications are violated

Avoid OOT in Future
Once the root cause of the OOT event has been established, determining corrective actions becomes imperative. Appropriate actions must be taken and documented to resolve the issue, prevent recurrence, and mitigate risks.
In some cases, amending the quality systems may be necessary as part of the corrective actions. This may involve revising specifications, procedures, training programs, or other elements to strengthen overall quality practices. Unexpected results often highlight opportunities for sustainable improvement.
Summary
Ensuring patient safety and product quality requires careful investigation of every unexpected result. Life science companies dedicated to sustaining life and well-being view deviations as valuable feedback, and a compliant response ensures that this feedback is heard. By rigorously following up on OOT events, these companies can maintain high standards, fulfill compliance obligations.
And foster a culture of quality through continuous learning and adjustment. Each unexpected signal represents a chance to refine understanding, strengthen patient and stakeholder trust, and transform unpredictability into process excellence. Behind every anomaly lies an answer and a key to forging the consistency that creates a world-class manufacturing system.

